GPCR activation monitoring: the GAPL biosensors
How the biosensors work
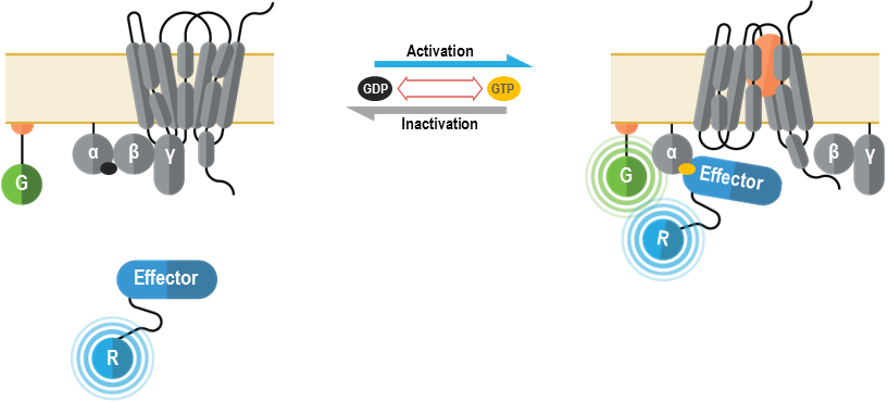
The Gα plasma membrane biosensors are used to monitor the activation of heterotrimeric G proteins at the plasma membrane upon receptor stimulation. Heterotrimeric G proteins are the canonical signaling partners of G protein-coupled receptors (GPCRs). Receptor activation triggers the exchange of a Gα-bound GDP for GTP, resulting in a conformational rearrangement of the heterotrimeric G protein that promotes dissociation of Gα and Gβ/γ subunits. In turn, GTP-bound Gα and free Gβ/γ subunits (β/γ remain associated) are then available to engage specific effectors. G proteins are grouped into families based on the signaling outcomes following activation of the Gα subunit. The Gs family (Gαs, Gαolf) bolsters the production of cAMP through direct activation of adenylyl cyclases (AC). Conversely, the Gi family (Gαi1, Gαi2, Gαi3, GαoA, GαoB and Gαz) reduces cAMP levels by inhibiting specific ACs. The Gq family (Gαq, Gα11, Gα14 and Gα15) activates Phospholipases Cβ (PLCβs) to produce the second messengers diacylglycerol (DAG) and inositol triphosphate (IP3), which subsequently promote the activation of PKCs and Ca2+ release from the endoplasmic reticulum, respectively. Finally, G12/13 family (Gα12 and Gα13) is known to control Rho-GEFs such as LARG, p115 and TRIO and thus influence processes linked to cytoskeletal remodeling (e.g., chemotaxis).
The bioSens-All® multimolecular GAPL BRET sensors were designed to monitor the plasma membrane recruitment of effector proteins that interact with active Gα subunits in a G protein family-selective manner. Such proteins/protein domains are fused at the N-terminus of Renilla luciferase (RLuc; R in following figure) and translocated to the plasma membrane upon GPCR activation. Membrane recruitment of these biosensors translates into an increased BRET efficiency with a plasma membrane anchored green fluorescent protein (GFP; G in figure below).
GAPL biosensor data generated with the Parathyroid Hormone 1 receptor
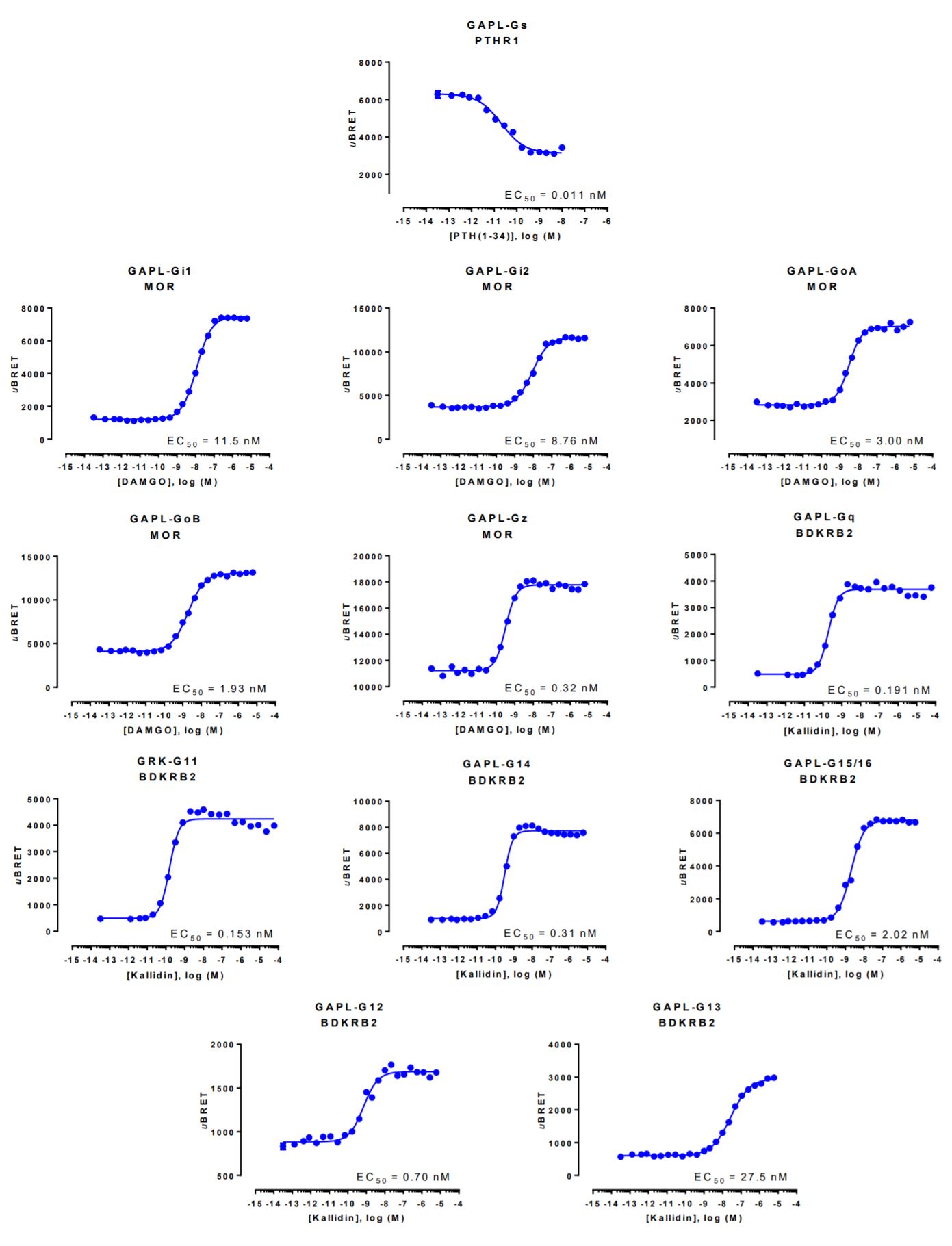
HEK293 cells were transfected with a receptor coding plasmid (either human parathyroid hormone type 1 receptor (PTHR1), human mu opioid receptor (MOR) or human bradykinin receptor B2 (BDKRB2)) in addition to plasmids coding for the GAPL biosensor. On the day of BRET, cells were rinsed with assay buffer, incubated with coelenterazine and increasing amounts of PTH(1-34), DAMGO or kallidin for 10 minutes and BRET subsequently measured.